Gene Therapy Analytical Development Summit – What Occured In 2023:
The 5thAnnual Gene Therapy Analytical Development Summit was the industry’s leading meeting to discover and discuss how the latest analytical tools are being developed and applied to guarantee the consistency, quality, and safety of gene therapy products.
Attendees in Boston were able to discover and learn the latest insights with case study presentations, informative panel discussions, and deep-dive workshops from Health Canada, Eli Lilly, Novartis Gene Therapies, Biogen, Spark Therapeutics, Astellas Gene Therapies, Bristol Myers Squibb, Biomarin Pharmaceuticals and many more!
Bridging scientist-level experts presenting the latest data and case studies direct from the lab, and senior decision makers navigating discussion through key analytical breakthroughs and strategy, attendees experienced and enjoyed in-depth and valuable conversations perfect for those new to the field and looking to further relationships with new and current colleagues.
What Was New for 2023?

Brand-New BioPharma Presenting Companies: With the largest speaker faculty to date in 2023, attendees heard from experts from Sarepta Therapeutics, Daiichi Sankyo, AskBio, Voyager Therapeutics, Bridge Bio Gene Therapy, Adverum Biotechnologies, and more showcasing their latest advancements in gene therapy analytics

Deep-Dive Regulatory Discussion Day: Clear communication with regulators is pivotal to understand analytical expectations for gene therapy products. This brand-new discussion day was led by 7 analytical and CMC regulatory leaders to improve understanding of the regulatory landscape, hear takeaways from interacting with regulatory bodies, and interact directly with Health Canada and ex-FDA representatives

Diverse AAV & Non-AAV Case Study Presentations: On top of the Non-AAV vector Focus Day, this year’s program included non-AAV content embedded in the main program from bluebird bio, CSL Behring, Expression Therapeutics, Northeastern University, Coriolis Pharma, Prime Medicine, and Refeyn



5 Key Benefits for Attendees:





Hearing the Regulatory Perspective & Industry-Regulator Interactions: Attendees learnt from and interacted directly with global gene therapy regulatory bodies and top industry leaders to understand regulatory views on analytical development and comparability, and learn key takeaways from recent regulator interactions with Health Canada, Standards Coordinating Body, and Eli Lilly
Developing & Validating Analytical Bioassays for Potency & Expression: Attendees learnt how to enhance development of bioassays to measure potency and expression across all stages of product development, setting up bioassay development for success, assay validation, platform approaches, and preparing for commercial stages with BridgeBio and Ultragenyx Pharmaceuticals
Quantitatively Characterizing Genome Regulation & Viral Titer Metrics: Attendees compared and contrasted the capabilities of different analytical methods to quantitively sequence and characterize genome integrity, and optimize genome titer measurements with Sanofi and Novartis Gene Therapies
Enhancing Methods to Understand Empty-Full Particles & Measure Physicochemical Properties: Attendees furthered their understanding of full, empty, and partial particles, viral host cell proteins, and gene therapy aggregates by enhancing characterization methods across chromatography, AUC, mass spectrometry and more with Biogen and Spark Therapeutics
Optimizing Product Quality, Process Development & Manufacturing: Attendees discovered strategies to ensure robust process development and product quality compliance, addressing upstream and downstream process development, scaling up manufacturing, risk assessments, and defining CQAs with Sarepta Therapeutics and Bristol Myers Squibb



Who Did You Meet?
The 5th Annual Gene Therapy Analytical Development Summit was solely dedicated to enhancing the quality and diversity of analytical tools for gene therapy products, attracting a niche audience of industry analytical experts to ignite thought-provoking discussions that lead to long-term, mutually beneficial relationships in a rapidly evolving field.
With 12+ hours of dedicated networking time, 4 streamlined tracks of content, 8 pre-conference workshops, and doubled post-conference discussion day content with the brand-new regulatory focus day, this was your must-attend conference reuniting biopharma analytical development, quality assurance, quality control, process development, CMC, and regulatory experts working across viral and non-viral vectors.
What Your Peers Have to Say:
“The conference provides information on all aspects of analytical development in the field of gene therapy and is a best place to meet key opinion leaders from the field.”
2022 attendee, NIB


“This is one of the best conferences I have ever attended as a scientist and vendor. The title is on point and extremely relevant. The audience was highly competent, motivated, and curious”
2022 attendee, Malvern Panalytical
Download the 2023 Full Event Guide for more information on:
- Expert Speaker Faculty
- Full Access to the 4-day Conference Agenda
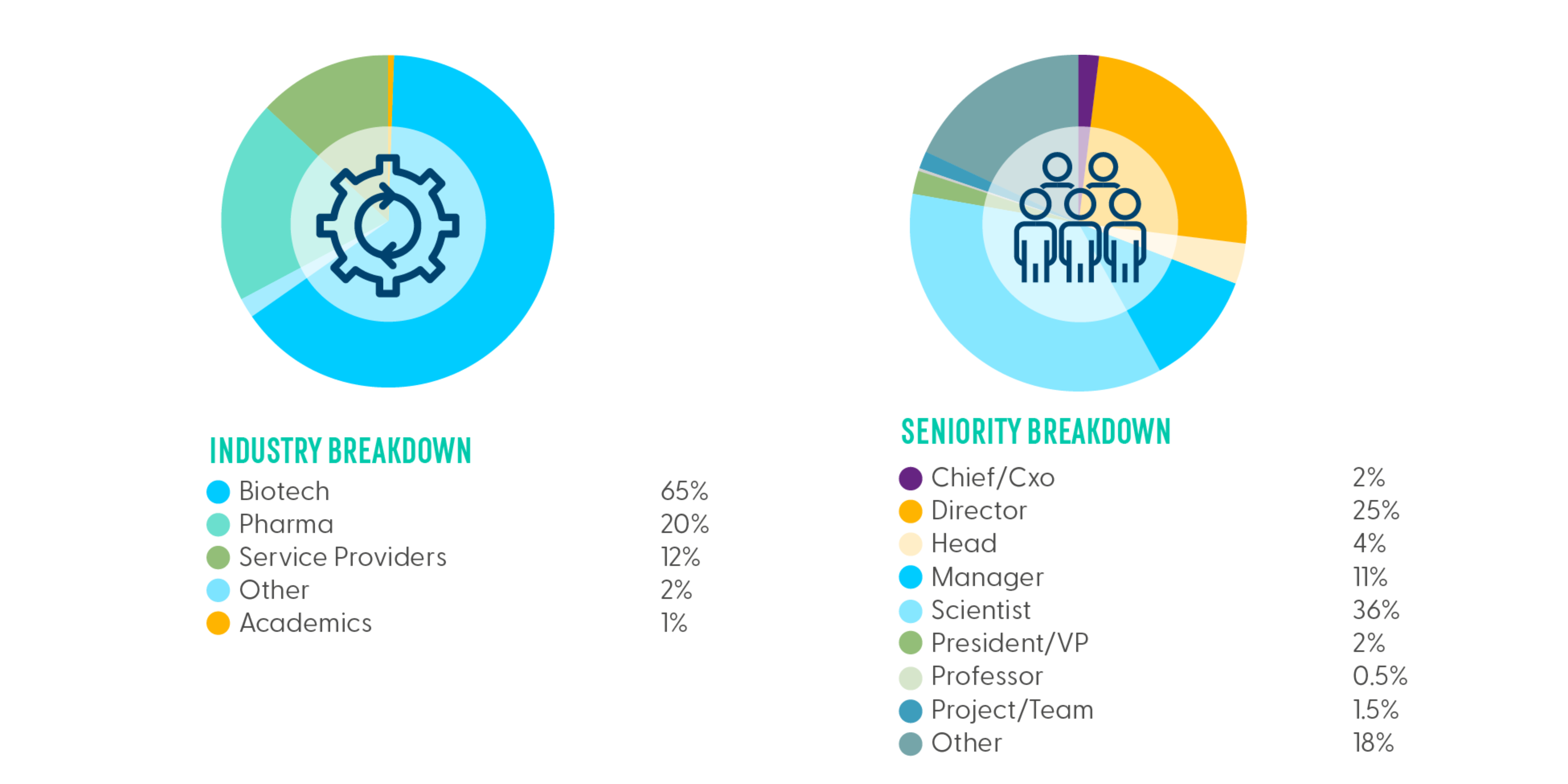
- Audience Breakdown
- & more

